-
Ultrafast Charge Recombination of Photogenerated Ion Pairs to an Electronic Excited State
A. Morandeira, L. Engeli and E. Vauthey
Journal of Physical Chemistry A, 106 (19) (2002), p4833-4837


DOI:10.1021/jp014393f | unige:3607 | Abstract | Article HTML | Article PDF
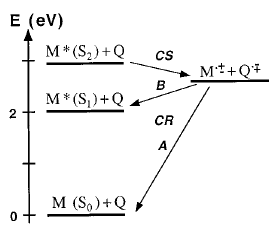
The dynamics of charge recombination (CR) of ion pairs formed upon electron-transfer quenching of Zn tetraphenylporphine (ZnTPP) in the S2Â state has been investigated by fluorescence upconversion. These ion pairs have two possible CR pathways:Â (A) a highly exergonic CR to the neutral ground state and (B) a moderately exergonic CR leading to the formation of ZnTPP in the S1Â state. Upon addition of quencher, the S2Â fluorescence decreases considerably, while the S1Â fluorescence is unaffected, indicating unambiguously that CR occurs via path B. A large fraction of the S2Â fluorescence quenching occurs in less than 100 fs. CR to the S1Â state of ZnTPP takes place with time constants around 400 fs.